The hydration temperature of low acyl gellan gum is very sensitive to the ionic environment and particularly sensitive to divalent ions. Low acyl gellan gum is a mixed salt and will only partially hydrate in cold deionized water. Gum hydration is further inhibited by the divalent ions in most water supplies. This inhibition makes low acyl gellan gum easy to disperse in cold water without forming lumps. Subsequently, the gum can be hydrated using sequestrants, heat or a combination of both
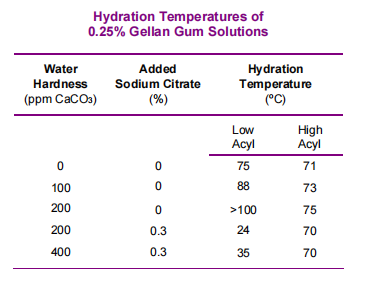
Calcium sequestrants, such as citrates and phosphates, can be used to control the divalent ions. Therefore, the hydration temperature of low acyl gellan gum can be effectively controlled. Without sequestrants, low acyl gellan gum requires a temperature of 75°C (167°F) to fully hydrate the gum. However, low acyl gellan gum can be hydrated in cold soft water using 0.3% sodium citrate, as seen in the chart below.
The setting temperature, melting temperature and final gel strength of low acyl gellan gum are also affected by cations and, therefore, sequestrants. Control of low acyl gellan gum properties can be achieved by balancing sequestrants, pH and added ions.
High acyl gellan gum will swell in deionized water creating a starch-like consistency. This swelling behavior can inhibited by low levels of sodium. The use of sodium to inhibit swelling is a useful strategy for improving gum dispersion and for minimizing viscosity during processing. Heat is required to fully hydrate high acyl gellan gum. While ions affect dispersion and particle swelling behavior, the hydration temperature of high acyl gellan gum is relatively insensitive to ions. High acyl gellan gum hydrates between 70°C and 80°C (158°F and 176°F) even in relatively high ion concentrations. Both forms of gellan gum can be dispersed directly in milk will hydrate during normal heat processing without sequestrants. Gum hydration is inhibited by soluble solids and low pH for both forms of gellan gum. In high solids systems, extra care must be taken to ensure that the gellan gum hydrates. In acidic environments, the pH must be above 4 for good hydration.
originally published at https://www.cinogel.com